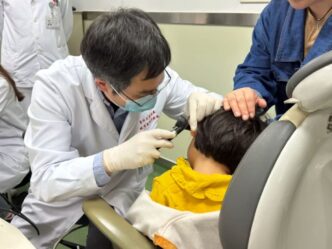
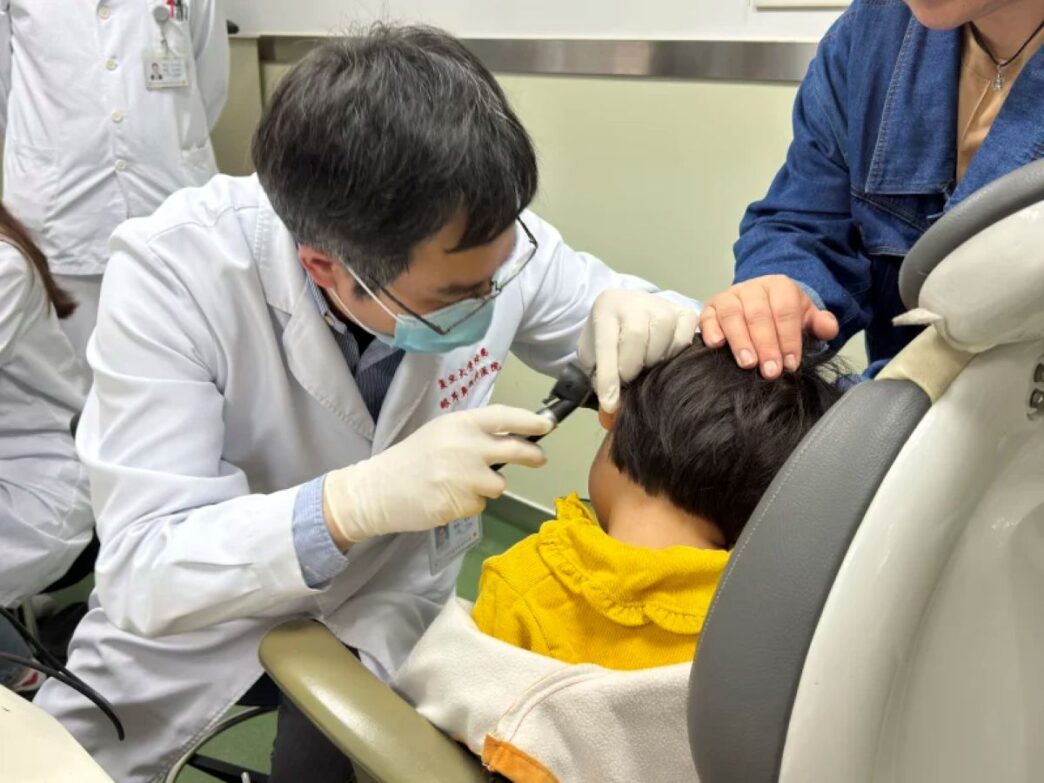
US health officials have approved a groundbreaking gene therapy designed to treat a rare inherited form of hearing loss, marking a major advance in modern medicine and raising hopes for future treatments targeting other forms of deafness.
The treatment, known as Otarmeni, was developed by American biotechnology company Regeneron and received approval from the US Food and Drug Administration on Thursday. It is the first therapy of its kind to address hearing loss caused by mutations in the OTOF gene, which plays a critical role in sending sound signals from the inner ear to the brain.
Health experts estimate that two to three out of every 1,000 children in the United States are born with hearing impairment. More than half of early-onset hearing loss cases are believed to be linked to genetic mutations. Otarmeni specifically targets a rare condition affecting around 50 newborns in the US each year.
The therapy is administered through a one-time surgical injection into the ear and will be available for children and adults suffering severe-to-profound hearing loss linked to the OTOF mutation.

Although gene therapies in the United States often cost millions of dollars per patient, Regeneron has announced that Otarmeni will be offered free of charge to eligible American patients.
The approval has already changed lives. Sierra Smith, whose young son Travis received the treatment during trials, described the results in emotional terms. “It’s absolutely incredible,” she said. Smith explained that before treatment, her son could not hear his own name or her voice telling him she loved him. Now, she said, “he can listen to music, and he loves it, and he loves to dance, and he loves instruments.”
Clinical trials involving 20 children aged between 10 months and 16 years produced strong results, with at least 80 per cent showing significant hearing improvement within months.
Dr Eliot Shearer, who worked on the trial, said the approval “signals a new era in the treatment of genetic forms of hearing loss, where reinstating 24/7 natural hearing is now possible.”
Shearer added that he had personally witnessed a child in the trial respond to their mother’s voice for the first time, underscoring the life-changing potential of the therapy.


 Trending
Trending