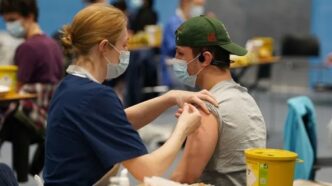
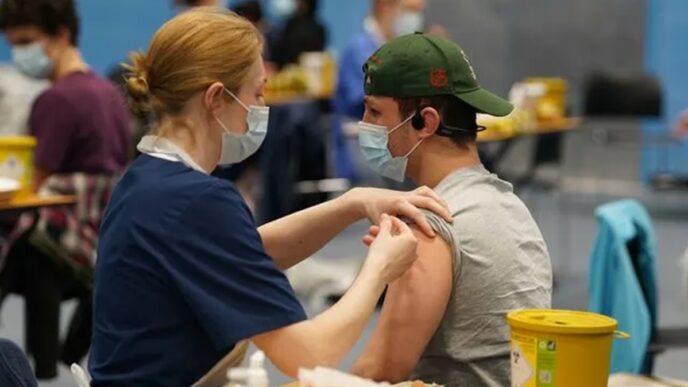
Japan has authorised pioneering stem-cell therapies for Parkinson’s disease and severe heart failure, marking a global milestone in regenerative medicine.
These treatments, which utilise induced pluripotent stem (iPS) cells, are expected to be commercially available to patients as early as this summer.
This achievement follows the Nobel Prize-winning research of Japanese scientist Shinya Yamanaka, who discovered how to return mature cells to a versatile, juvenile state.
Sumitomo Pharma received approval for Amchepry, a treatment that transplants iPS-derived precursors of dopamine-producing cells into the brains of Parkinson’s patients.
Clinical trials involving seven participants indicated that the therapy is safe and effective in improving motor symptoms without major adverse effects.

Additionally, the health ministry approved ReHeart, a medical sheet developed by Cuorips that helps restore heart function by forming new blood vessels in damaged heart muscle.
To expedite patient access, the treatments were granted “conditional and time-limited” approval, a provisional licensing system used when safety and efficacy are demonstrated in smaller patient groups.
Health Minister Kenichiro Ueno expressed hope that these ground-breaking therapies will eventually provide relief to millions of patients suffering from these chronic conditions worldwide.


 Trending
Trending